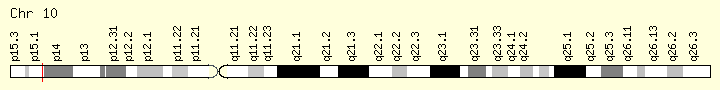What is Latent Autoimmune Diabetes in Adults (LADA)?
|
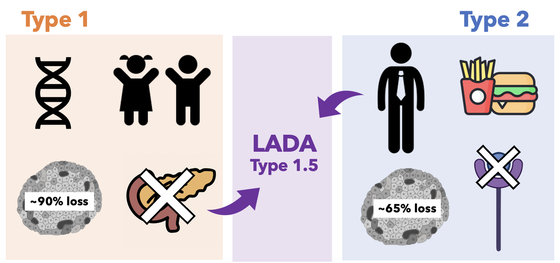
Latent Autoimmune Diabetes in Adults, or LADA, is a slowly progressive form of Type 1 diabetes. It is characterized by the presence of pancreatic autoantibodies and initial insulin independence. Unlike Type 1 diabetes, LADA is usually diagnosed in people that are between 30-50 years old, so it can easily be misdiagnosed as Type 2 diabetes. Because of this misdiagnoses, LADA is also under diagnosed due to the lack of autoantibody screenings. However, LADA is a different disease from Type 1 or Type 2 diabetes. [1]
LADA is known to be more heterogeneous than Type 1 diabetes, which means that there are multiple set of causes for the disease. This includes the variability in beta-cell destruction, insulin resistance, and pattern of islet autoantibodies. [3] |
|
Figure 1. The general difference between T1D, T2D, and LADA. Like T1D, LADA has autoantibodies that destroys beta-cells in the pancreas, but it's age of onset is 35 and above. Thus, the destruction of beta-cells are slower in LADA than in T1D [1]. Because LADA has characteristics of both T1D and T2D, it is also known as Type 1.5 diabetes.
|
What are the symptoms of LADA?
|
The initial symptoms of LADA include feeling tired after meals, foggy headedness, and the feeling of hunger even after meals. As the individual's ability to produce insulin decrease through out time, symptoms such as excessive thirst, frequent urination, and blurred vision can occur. [2] The symptoms are similar to that of Type 2 diabetes, which makes it much easier for individuals with LADA to get a misdiagnosis. In fact, it is known that 5 to 10% of Type 2 diabetes diagnoses are actually LADA. [1]
|
|
Figure 2. LADA is easily misdiagnosed as T2D because of its age of onset and similarity of symptoms.
|
How do we test and treat for LADA?
|
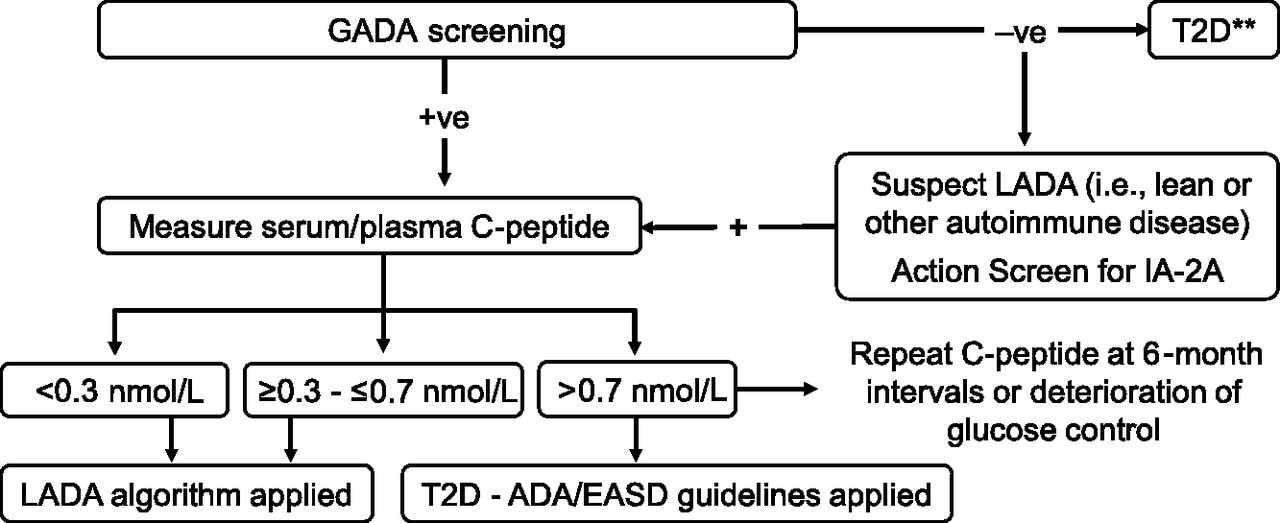
Figure 3. An algorithm for diagnosing LADA based on GADA screening and measuring C-peptide levels. Adapted from Pozzilli et al. 2018.
|
LADA is diagnosed through the combination of the age of onset, presence of at least one circulating islet autoantibody, and lack of insulin 6 months after diagnosis. The age of onset is generally more than 30 years of age. The accumulation of autoantibodies are also an important biomarker in diagnosing LADA, however, GADA screenings are known to be the best single marker as other autoantibodies are reported to be infrequent.
Unfortunately, there are no specific guidelines for therapy, nor is there an established treatment algorithm after diagnoses of LADA and is most likely treated as patients with Type 2 diabetes. [4] However, the heterogeneity of LADA suggests a more personalized approach to treatment. For example, LADA could be treated differently by the variation of C-peptide levels [9]. |
PFKFB3 and LADA
Figure 4. The PFKFB3 gene is located on chromosome 10.
The PFKFB3 gene encodes the protein 6-Phosphofructo-2-Kinase/Fructose-2,6-Biphosphatase 3 which is involved in regulation of glycolysis in eukaryotes. This protein is required for cell cycle progression and prevention of apoptosis, regulating glucose metabolism as well as cell proliferation[5]. Because of its role in glycolysis regulation, PFKFB3 is closely related to tumor cells as well. PFKFB3 controls glycolytic flux which supports proliferation of cancer cells through providing energy by glycolysis. Its inhibition has been reported to suppress growth of tumors, increasing the possibility of using PFKFB3 inhibitors as a therapeutic strategy against cancer[6].
Interestingly, studies show that PFKFB3 knockouts in animal studies showed systemic insulin resistance and involvement in inflammation. In patients with rheumatoid arthritis, the loss of PFKFB3 in T cells has shown to increase cellular apoptosis and decreased immune response [7]. A GWAS study between LADA, Type 1 diabetes, and Type 2 diabetes showed that PFKFB3 may be the factor that distinguished child-onset Type 1 diabetes from LADA and the most likely functional gene candidate for LADA [8].
Interestingly, studies show that PFKFB3 knockouts in animal studies showed systemic insulin resistance and involvement in inflammation. In patients with rheumatoid arthritis, the loss of PFKFB3 in T cells has shown to increase cellular apoptosis and decreased immune response [7]. A GWAS study between LADA, Type 1 diabetes, and Type 2 diabetes showed that PFKFB3 may be the factor that distinguished child-onset Type 1 diabetes from LADA and the most likely functional gene candidate for LADA [8].
|
Figure 5. GWAS of LADA and T1D. PFKFB3 was found to be the most likely functional gene that distinguishes LADA and T1D. Adapted from Cousminer et al., 2018.
|
|
Figure 6. Activated HIF1a binds to PFKFB3 and upregulates transcription, leading to an increase in cell viability. This pathway is activated during the slow beta-cell loss phase of both pre-T1D and T2D [10]. This image was created using biorender.com
|
Organizations
References
Header image: https://www.diabetes.co.uk/Diabetes-and-Hyperglycaemia.html
1. Pozzilli, P., & Pieralice, S. (2018). Latent Autoimmune Diabetes in Adults: Current Status and New Horizons. Endocrinology and metabolism (Seoul, Korea), 33(2), 147–159. https://doi.org/10.3803/EnM.2018.33.2.147
2. Diabetes Digital Media Ltd. Diabetes.co.uk: the global diabetes community website. January 15th 2019. Accessed February 20th 2021.
3. Mishra, R., Hodge, K. M., Cousminer, D. L., Leslie, R. D., & Grant, S. (2018). A Global Perspective of Latent Autoimmune Diabetes in Adults. Trends in endocrinology and metabolism: TEM, 29(9), 638–650. https://doi.org/10.1016/j.tem.2018.07.001
4. Cernea, S., Buzzetti, R., & Pozzilli, P. (2009). Beta-cell protection and therapy for latent autoimmune diabetes in adults. Diabetes care, 32 Suppl 2(Suppl 2), S246–S252. https://doi.org/10.2337/dc09-S317
5. https://www.genecards.org/cgi-bin/carddisp.pl?gene=PFKFB3
6. Lu, L., Chen, Y., & Zhu, Y. (2017). The molecular basis of targeting PFKFB3 as a therapeutic strategy against cancer. Oncotarget, 8(37), 62793–62802. https://doi.org/10.18632/oncotarget.19513
7. Koufakis, T., Karras, S. N., Zebekakis, P., & Kotsa, K. (2018). Results of the First Genome-Wide Association Study of Latent Autoimmune Diabetes in Adults further highlight the need for a novel diabetes classification system. Annals of translational medicine, 6(Suppl 2), S102. https://doi.org/10.21037/atm.2018.11.40
8. Cousminer, D. L., Ahlqvist, E., Mishra, R., Andersen, M. K., Chesi, A., Hawa, M. I., Davis, A., Hodge, K. M., Bradfield, J. P., Zhou, K., Guy, V. C., Åkerlund, M., Wod, M., Fritsche, L. G., Vestergaard, H., Snyder, J., Højlund, K., Linneberg, A., Käräjämäki, A., Brandslund, I., … Grant, S. (2018). First Genome-Wide Association Study of Latent Autoimmune Diabetes in Adults Reveals Novel Insights Linking Immune and Metabolic Diabetes. Diabetes care, 41(11), 2396–2403. https://doi.org/10.2337/dc18-1032
9.Buzzetti, R., Tuomi, T., Mauricio, D., Pietropaolo, M., Zhou, Z., Pozzilli, P., & Leslie, R. D. (2020). Management of Latent Autoimmune Diabetes in Adults: A Consensus Statement From an International Expert Panel. Diabetes, 69(10), 2037–2047. https://doi.org/10.2337/dbi20-0017
10. Nomoto H, Pei L, Montemurro C, et al. Activation of the HIF1α/PFKFB3 stress response pathway in beta cells in type 1 diabetes. Diabetologia. 2020;63(1):149-161. doi:10.1007/s00125-019-05030-5
Figure 3: Buzzetti R, Tuomi T, Mauricio D, et al. Management of Latent Autoimmune Diabetes in Adults: A Consensus Statement From an International Expert Panel. Diabetes. 2020;69(10):2037-2047. doi:10.2337/dbi20-0017
Figure 4: https://www.genecards.org/cgi-bin/carddisp.pl?gene=PFKFB3
Figure 5: Cousminer, D. L., Ahlqvist, E., Mishra, R., Andersen, M. K., Chesi, A., Hawa, M. I., Davis, A., Hodge, K. M., Bradfield, J. P., Zhou, K., Guy, V. C., Åkerlund, M., Wod, M., Fritsche, L. G., Vestergaard, H., Snyder, J., Højlund, K., Linneberg, A., Käräjämäki, A., Brandslund, I., … Grant, S. (2018). First Genome-Wide Association Study of Latent Autoimmune Diabetes in Adults Reveals Novel Insights Linking Immune and Metabolic Diabetes. Diabetes care, 41(11), 2396–2403. https://doi.org/10.2337/dc18-1032 (adapted)
Figure 6: Created with biorender.com
Footer image: linked